
The pI of amino acids with acidic side chains For example, aspartic acid has a pK a3 corresponding to its CH 2CO 2H side chain. Subsequently, the neutral form arises under conditions of acidity, when the additional -1 charge has been neutralized. 
In cases where the side chain is acidic, the pI is at a lower pH because the acidic side chain will result in an additional -1 charge. More complex amino acids have more than two pK a values due to the presence of additional pK a values for their side chains. The isoelectric point can, therefore, be approximated by averaging the two pK a values. If the pH s increased to that of the pK a of the amino group (9.60), another buffer is produced where there is an equilibration between the protonated neutral zwitterion and the deprotonated negative amino acid. This pH produces the carboxyl buffer zone. For example, when the pH = 2.34 (pK a of glycine), the solution comprises of 50% neutral molecules in which the carboxyl is deprotonated, and 50% positive molecules where the carboxyl is protonated. 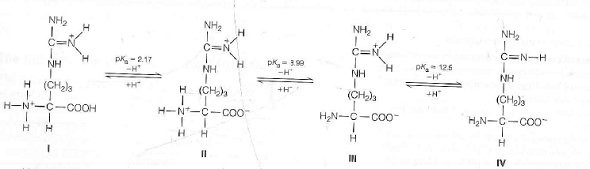 
When the pH is exactly at the pK a value, a buffer arises in which the deprotonated and protonated amino acids exist in equilibrium. How does the zwitterionic form of an amino acid relate to the pI? The zwitterionic form of the amino acid can exist at any pH value between pK a1 and pK a2. The amino acid now carries a net negative charge of -1. When the pH> pK a2, the amino group is deprotonated and loses its charge. This represents the zwitterionic form of the amino acid as it simultaneously possesses a positive and negative charge. When the pHpH> pK a1), the carboxyl group is deprotonated while the amino group remains protonated. Bariatric surgery associated with long-term improvements in pain, physical function, work productivity.Air pollution may trigger arrhythmias in healthy adolescents.Disruption of actin cytoskeleton is key to triggering human immune response, research finds.This is illustrated below using glycine, which has two pK a values – pK a 1 represents the (de)protonation of the carboxyl group and pK a2 represents the (de)protonation of the amine group: For example, if there were two pK a values, three structures can be discerned. There is always one more structure than the number of pK a values for amino acids. Amino acids possess distinct pK as for the carbonyl group, amino group, and any functional groups on the side chain that may be protonated or deprotonated. PK a relates to the equilibrium constant and defines the transition between two structural forms of an amino acid–protonated and deprotonated. The net charge of the protein is determined by summing the charge of individual amino acids across the protein. The pK a for an amino acid is an acid dissociation constant that refers to the equilibrium between the protonated and deprotonated forms of the backbone amino group, backbone carboxyl group, and any potential acid/base component of the variable group. What is the relationship between the pH and pK a of an amino acid? A zwitterionic amino acid consists of a negative carboxylate ion and a positive ammonium ion alongside a charge on their side chain, which varies between amino acids. Zwitterion possesses discrete functional groups, each of which carry a positive or negative charge. The isoelectric point corresponds to what is known as the zwitterionic form of a protein. This charge is also dependent on the pH of the surrounding solution. Each of the amino acids in a protein carries a distinct charge, and the overall charge of a protein is the summation of the individual charges on each amino acid. pI is most commonly examined for proteins. The isoelectric point (pI) is the point at which the net charge on a molecule is zero.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
